EHDV PCR Kit
Bio-T kit® EHDV all genotypes
The EHDV PCR kit is a ready-to-use kit for the detection of all strains of EHDV (Epizootic Hemorrhagic Disease Virus) by real-time PCR for the diagnosis of Epizootic Hemorrhagic Disease (EHD) in ruminants. This EHDV PCR test contains an endogenous IPC internal positive control. It has been validated by the French National Reference Laboratory for FCO (Fièvre Catharrale Ovine) (ANSES, Maisons-Alfort).
✔ EHDV duplex PCR kit developed in compliance with French standard U47-600-2 (AFNOR) and validated by the French FCO NRL (ANSES, Maisons-Alfort)
✔ Rapid defrosting of the ready-to-use, blue-colored Master Mix for easy distinction
✔ Reliable detection of EHDV by real-time PCR thanks to an endogenous IPC to ensure high-quality results on all matrices, even the most difficult ones
✔ Sensitivity & specificity of Epizootic Haemorrhagic Disease diagnosis with our kits: 100% on blood and spleen matrices
✔ Matrices: Whole blood (on EDTA tubes) and organ (spleen). Individual analyses.
✔ Kit developed on AriaMx™ (Agilent Technologies, Fast ramping by default) and confirmed on ABI PRISM® 7500 Fast (Applied Biosystems) in standard ramping, on QuantStudio™ 5 (Applied BioSystems) in standard ramping, on Light Cycler® 480 (Roche Diagnostics) in standard ramping, on CFX96™ (BioRad) in standard ramping and on MIC qPCR cycler (Bio molecular systems) in fast ramping by default. It is compatible with all thermal cyclers with at least 6-FAM and Cy5 reading channels. For further information, please contact our technical support.
✔ Kit validated on Mic qPCR cycler (Bio molecular systems) with 60 min run
✔ Extraction and amplification program common to all kits for Ruminant species
✔ Companion product : EHDV quantified RNA (cARN-EHDV-001)
✔ French manufacturing
- Method
- qRT-PCR
- Species
- Ruminants
- Pathogen
- Epizootic haemorrhagic disease virus
- Brand
- Biosellal
You must be logged to access the documents
Log in to your customer accountAbout Epizootic Haemorrhagic Disease
Epizootic Hemorrhagic Disease (EHD), also known as viral hemorrhagic fever, is a serious viral infection affecting ruminants, including cattle, sheep and goats. The disease is caused by a virus of the Bunyaviridae family, transmitted mainly by ticks of the genus Ixodes. Symptoms of MHE include fever, internal and external bleeding, neurological disorders, and in the most severe cases, death.


Diagnosis & prevention of Epizootic Haemorrhagic Disease
The disease can cause significant economic losses for livestock farmers, due to high mortality in infected animals and trade restrictions imposed to prevent the spread of the disease. Early diagnosis of Epizootic Haemorrhagic Disease (EHD) is crucial to the implementation of effective control measures. EHDV PCR kits such as the one developed by BioSellal offer a rapid and accurate solution for detecting the EHDV virus in animal samples, enabling farmers and veterinarians to intervene rapidly to limit the spread of the disease and protect their herds.
Made in France
Opt for quality and local know-how.
Iso-certified
A guarantee of quality in line with international standards.
Secure payment
Your transactions are completely secure.
Availability & support
Our technical support team is on hand to help you with your analyses.
Frequently asked questions
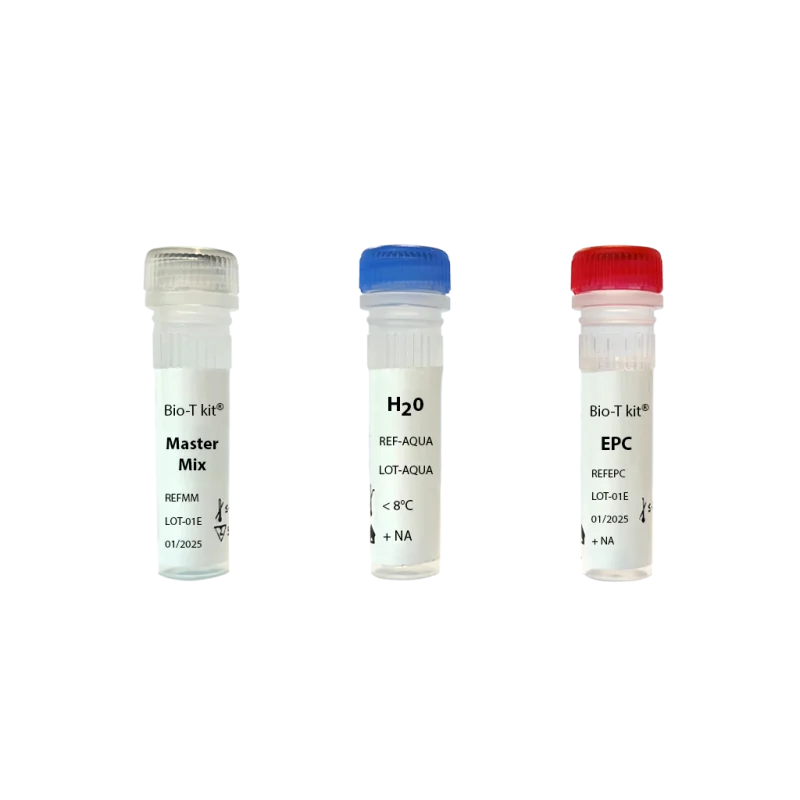
Each real-time PCR kit includes :
- Ready-to-use Master Mix (blue color)
- Positive amplification control, containing targeted pathogens - EPC
- Negative amplification control - water.
- Exogenous IPC (Internal positive control)
Each ELISA kit includes at least :
- Test plate with 12 x 8-well strips
- Sample diluent
- Negative control
- Positive control
- 100X or ready-to-use conjugate
- Conjugate diluent
- Wash solution
- Substrate
- Stop solution
- Plate adhesives
Each product comes with a certificate of analysis and detailed instructions for use to ensure optimum use and reliable results.
To ensure the integrity and efficiency of your kits, follow these storage recommendations:
Real-time PCR kits:
On receipt: Store kits at a temperature ≤ -16°C. This preserves the quality and performance of the kit components.
ELISA kits:
On receipt: Store components at 5 ± 3°C to maintain efficacy. The concentrated wash solution can be stored at room temperature, i.e. 21 ± 4°C.
By respecting these storage conditions, you ensure the reliability and optimum performance of your kits for accurate and consistent results.
- The shelf life of a PCR kit for the detection of RNA pathogens (viruses) is 12 months.
- The shelf-life of a PCR kit for the detection of DNA pathogens (viruses, bacteria, parasites) is 24 months.
- The shelf-life of an ELISA kit is 15 or 18 months.
At BioSellal, we are constantly seeking to improve our products. We regularly carry out stability studies to extend expiration dates, guaranteeing that our kits will always perform better and last longer.
Yes, absolutely! At BioSellal, we're committed to providing our customers with complete solutions to carry out their analyses under optimum conditions. That's why, for the majority of our PCR and ELISA kits, we offer in-house reference materials, guaranteeing you reliable, accurate results every time.
The quality management system implemented by BioSellal was certified ISO 9001 (version 2015) by AFNOR in June 2017 and has since been renewed for the following areas: Research and development activities, production, industrialization and sales and technical support of PCR and ELISA kits for veterinary diagnostics. BioSellal carries out several levels of quality control on product batches, as well as validation of raw materials.
All PCR kits are developed and validated to NF U47 600-2 standard. All ELISA kits are developed and validated in accordance with standards NF U47-310, NF U47-301, NF U47-300, NF U47-020, NF U47-019.
Yes, that's right! Our PCR kits are designed to be compatible with all open devices with FAM, VIC/HEX, CY5 and Texas Red/ROX reading channels. So you can use them with confidence on a wide range of thermal cyclers for optimum results.
At BioSellal, we have a particular preference for the MIC thermal cycler, the most compact, accurate and fast thermal cycler on the market.
Our various departments are always ready to answer your questions and help you. Here's how to contact us:
Sales Administration & Logistics (for all questions concerning delivery, lead times, expiry dates, batches, etc.)
Mail: contact@biosellal.com
- Phone: +33 (0)4 26 78 32 48
Sales Department (for all questions regarding information on a product, a quote, a partnership, etc.)
- Mail: commercial@biosellal.com
- Phone: +33 (0)4 26 78 32 57
Technical Support (for technical assistance on our products or for all questions following analyses)
- Mail: tech@biosellal.com
- Tel: +33 (0)4 26 78 32 48
Our dedicated teams are here to ensure you have the best possible experience with our products. We look forward to hearing from you!
Absolutely! At BioSellal, we offer customized training to maximize the use of our technologies, including real-time PCR, ELISA, sequencing and digital PCR. Whether you want to discover our products or deepen your knowledge, we're here to support you every step of the way.
All documents relating to our products are available directly on our website. Simply create an account and log in for easy access to all our documentation resources.